If one atom is more electronegative, the electrons of the bond are more attracted to that atom. If one atom is overwhelmingly more electronegative than the other atom, the electrons will not be shared and an ionic bond will result.
Is electronegativity a force of attraction?
Electronegativity is the ability to attract electrons to an atom. This attraction is electrostatic in nature. It is the attraction between the nucleus (positive) and the valence electrons that is crucial in determining the electronegativity of the atom.
What is the force of attraction between two atoms called?
Chemical bond refers to the forces holding atoms together to form molecules and solids. This force is of an electric nature, and the attraction between electrons of one atom to the nucleus of another atom contributes to what is known as chemical bonds.
How does electronegativity affect bonding between two atoms?
Electronegativity differences affect the degree of sharing in covalent bonding. The more equal the sharing the stronger the bond. If the electronegativities of the two atoms are completely the same, the bond formed by the sharing of the electrons will be a pure covalent bond.
How does a difference in electronegativity between two atoms lead to a polar bond or molecule?
The shared electrons of the covalent bond are held more tightly at the more electronegative element creating a partial negative charge, while the less electronegative element has a partial positive charge, . The larger the difference in electronegativity between the two atoms, the more polar the bond.
How does electronegativity affect intermolecular forces?
The difference in electronegativity between the two atoms in a bond determines the type of bond that is formed. Intermolecular forces, such as dipole-dipole forces, London dispersion forces, and hydrogen bonds, are weak forces that exist between molecules.
How do you know the electronegativity of an atom?
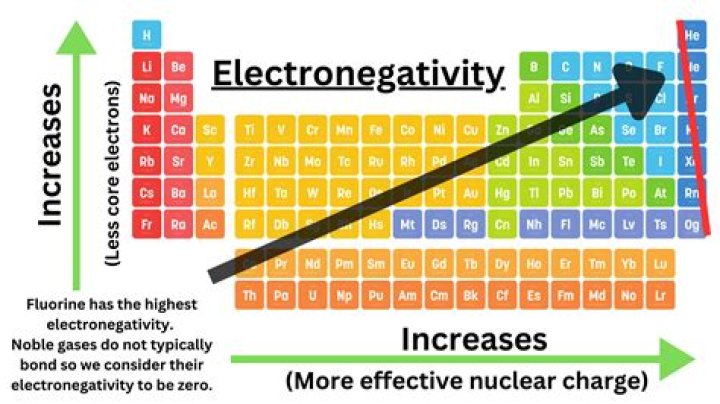
On the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. As a result, the most electronegative elements are found on the top right of the periodic table, while the least electronegative elements are found on the bottom left.
What force allows atoms to bond?
Chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction.
What is a electrostatic attraction?
Electrostatic attraction is essentially the attraction between the positively charged nucleus of an atom and the negatively charged electrons surrounding it. The greater the positive charge of the nucleus (the more protons), the greater the effective nuclear charge.
How does electronegativity relate to polar and nonpolar covalent bonds?
Electrons in a polar covalent bond are shifted toward the more electronegative atom; thus, the more electronegative atom is the one with the partial negative charge. The greater the difference in electronegativity, the more polarized the electron distribution and the larger the partial charges of the atoms.
How does the electronegativity of bonding atoms determine the kind of bond formed between them?
Electronegativity is a measure of the tendency of an atom to attract electrons (or electron density) towards itself. It determines how the shared electrons are distributed between the two atoms in a bond. The more strongly an atom attracts the electrons in its bonds, the larger its electronegativity.