Vomiting or nasogastric (NG) suction generates metabolic alkalosis by the loss of gastric secretions, which are rich in hydrochloric acid (HCl). Whenever a hydrogen ion is excreted, a bicarbonate ion is gained in the extracellular space.
Why is metabolic alkalosis a potential complication of vomiting?
In metabolic alkalosis there is excess of bicarbonate in the body fluids. It can occur in a variety of conditions. It may be due to digestive issues, like repeated vomiting, that disrupt the blood’s acid-base balance.
Can normal saline be used for metabolic alkalosis?
For severe metabolic alkalosis, don’t rely on a single treatment (e.g., normal saline). Rather, a multimodal strategy may be most effective, with attention to all factors which may be perpetuating the metabolic alkalosis.
Why does vomiting cause metabolic acidosis?
Vomiting causes loss of hydrogen and chloride ions, and thereby allows the stomach to add new bicarbonate to the body (Figure 1). As a primary abnormality, this may be detrimental, causing ‘gastric alkalosis’ [4. The response of normal man to selective depletion of hydrochloric acid.
What is metabolic alkalosis and explain how excessive vomiting can change blood pH?
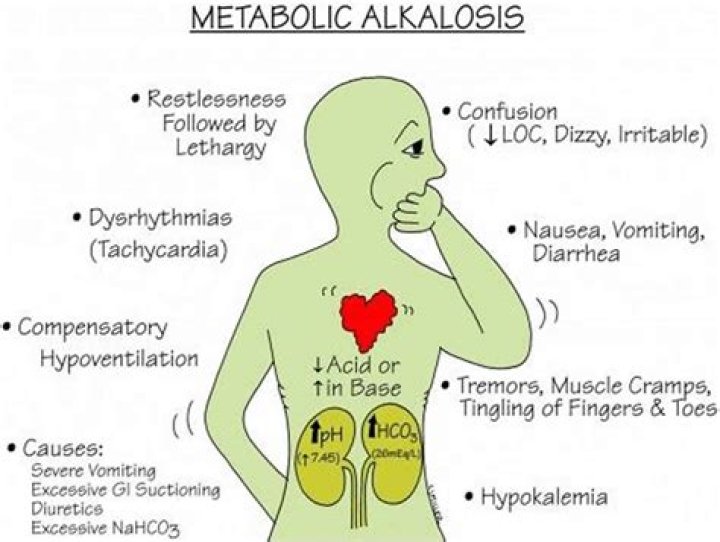
Metabolic alkalosis is primary increase in bicarbonate (HCO 3 −) with or without compensatory increase in carbon dioxide partial pressure (Pco 2); pH may be high or nearly normal. Common causes include prolonged vomiting, hypovolemia, diuretic use, and hypokalemia.
How can prolonged vomiting affect the pH?
Expelling the gastric acid contents causes the loss of chloride and hydrogen ions which can lead to hypochloremic metabolic alkalosis, where there are high levels of bicarbonate and carbon dioxide but low levels of chloride, leading to an increased blood pH.
Why does hypokalemia cause vomiting?
Vomiting leads to hypokalemia via a complex pathogenesis. Gastric fluid itself contains little potassium, approximately 10 mEq/L. However, vomiting produces volume depletion and metabolic alkalosis, which are accompanied by increased renal potassium excretion.
What does vomiting do to blood pH?
Your blood pH levels will test normal, however your kidneys are releasing more bicarbonate, compensating for the lower levels of carbon dioxide. When your blood has too much bicarbonate, it is called metabolic alkalosis. This can happen from prolonged vomiting.
What are the signs and symptoms of saline-responsive alkalosis?
Saline-responsive alkalosis is characterized by normotensive extracellular volume contraction (contraction alkalosis) and hypokalemia. Hypotension and orthostasis may be seen.
What are the different types of metmetabolic alkalosis?
Metabolic alkalosis is divided into three categories: sodium chloride–responsive metabolic alkalosis, sodium chloride–resistant metabolic alkalosis]
What causes chloride-responsive metabolic alkalosis?
The most common causes are volume depletion (particularly when involving loss of gastric acid and chloride (Cl) due to recurrent vomiting or nasogastric suction) and diuretic use. Metabolic alkalosis involving loss or excess secretion of Cl is termed chloride-responsive.
How do you evaluate for compensatory metabolic alkalosis?
VBG/ABG to evaluate for compensatory metabolic alkalosis. Urine potassium <20-30 mM suggests that hypokalemia may be contributory. Chloride <10-30 mM suggests saline responsive. Chloride >10-30 mM suggests saline unresponsive. Generally, this isn’t useful in critical care.