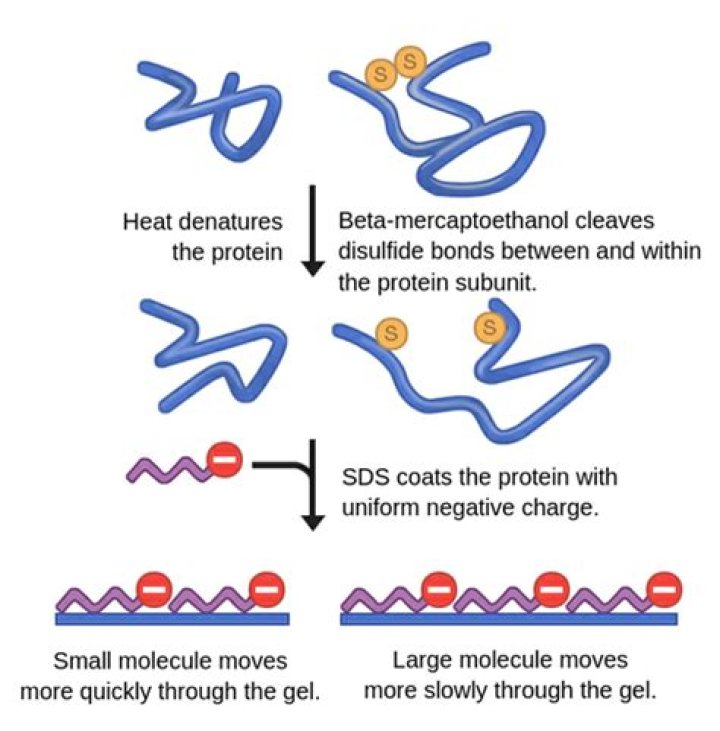
SDS imparts uniform negative charge and linearises your protein and Beta-mercaptoethanol breaks cysteine-cysteine disulphide bridges. Heating your protein containing SDS and Beta-mercaptoethanol helps denature the protein. Heating speeds up this breakdown process and the amount of heating is to be optimized in the lab.
Why is there usually B mercaptoethanol in the sample buffer?
Why do we need add beta-mercaptoethanol in sample buffer to determine Bromelain’s MW in SDS-PAGE? The role of beta-mercaptoethanol is to break all the disulfide bonds and denature the protein of interest.
How much mercaptoethanol is in a sample buffer?
Add 50 µl of β-mercaptoethanol per 950 µl of sample buffer for a final concentration of 5% β-mercaptoethanol, 710 mM. As an alternative, dithiothreitol (DTT or Cleland’s reagent) may be used at a final concentration of 350 mM (54 mg/ml). Dilute 1 part sample with 1 part Laemmli sample buffer.
What does beta-mercaptoethanol do in SDS-PAGE?
BME is suitable for reducing protein disulfide bonds prior to polyacrylamide gel electrophoresis and is usually included in a sample buffer for SDS-PAGE at a concentration of 5%. Cleaving intermolecular (between subunits) disulfide bonds allows the subunits of a protein to separate independently on SDS-PAGE.
What is the function of beta mercaptoethanol?
Beta-mercaptoethanol (ß-ME) is a reducing agent that will irreversibly denature RNases by reducing disulfide bonds and destroying the native conformation required for enzyme functionality.
Why Tris buffer is used in SDS-PAGE?
Tris is the buffer used for most SDS-PAGE. Its pKa of 8.1 makes it an excellent buffer in the 7-9 pH range. SDS in the buffer helps keep the proteins linear. Glycine is an amino acid whose charge state plays a big role in the stacking gel.
What does B mercaptoethanol do?
How long does beta-mercaptoethanol last in buffer?
No. Beta-Mercaptoethanol (ß-ME) is stable for 1 month, but Buffer RLT itself is stable for at least 9 months at room temperature (15 to 25°C).
Is 2-mercaptoethanol the same as beta-mercaptoethanol?
Gibco™ 2-Mercaptoethanol (also known as beta-mercaptoethanol or BME) is a potent reducing agent used in cell culture media to prevent toxic levels of oxygen radicals.
What is the function of APS?
Ammonium persulfate (APS) is an oxidizing agent that is often used with tetramethylethylenediamine (TEMED, Part No. 17919) to catalyze the polymerization of acrylamide and bisacrylamide to prepare polyacrylamide gels for electrophoresis.
How do you neutralize beta mercaptoethanol?
BME odor can be neutralized using standard household bleach. Bleach acts as an oxidizer and converts the thiol group of beta mercaptoethanol into a sulfonic acid derivative which eliminates the natural gas odor. Be sure to absorb any excess BME liquid with an inert absorbent prior to odor decontamination with bleach.
How do you dilute beta-mercaptoethanol for SDS?
Dilute the 10x loading buffer 1:9 in your sample. Dilute β-mercaptoethanol 1:19 in your sample (i.e. 5% final concentration). Heath samples for 10 minutes at 95°C. Load on SDS-PAGE and run.
What is the function of ββ-mercaptoethanol and glycerol in runnig buffer?
β-mercaptoethanol is used to break disulphide bonds. Glycerol increases the density of the sample relative to the surrounding runnig buffer making it easier to load in the well. Bromophenol blue is used to follow the run of protein sample on the gel.
What is the use of BME buffer in SDS PAGE?
BME is suitable for reducing protein disulfide bonds prior to polyacrylamide gel electrophoresis and is usually included in a sample buffer for SDS-PAGE at a concentration of 5%. Cleaving intermolecular (between subunits) disulfide bonds allows the subunits of a protein to separate independently on SDS-PAGE.
What does beta mercaptoethanol do to proteins?
What does beta mercaptoethanol do to proteins? What does beta mercaptoethanol do to proteins? Beta-mercaptoethanol (ß-ME) is a reducing agent that will irreversibly denature RNases by reducing disulfide bonds and destroying the native conformation required for enzyme functionality.