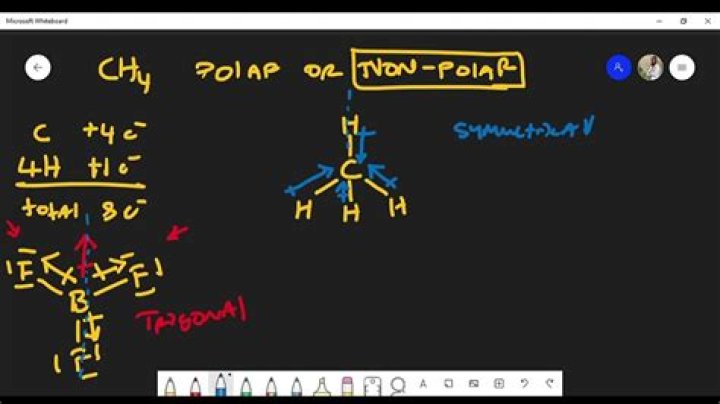
All the outer atoms are the same – the same dipoles, and that the dipole moments are in the same direction – towards the carbon atom, the overall molecule becomes non-polar. Therefore, methane has non-polar bonds, and is non-polar overall.
Is CH2I2 polar?
Using VSEPR, this molecule would have a tetrahedral shape. Even though tetrahedral is a symmetrical molecular shape, drawing out the dipole moments (from partial positive C atom to partial negative I atoms) shows that the dipoles do not cancel, thus making CH2I2 polar.
Does CH4 form a polar covalent bond?
2 Answers By Expert Tutors. Methane contains nonpolar covalent bonds. This is because there is a very small difference in the electronegativity of Carbon and Hydrogen. Thus they are going to share their electrons fairly equally.
Is dibromomethane a nonpolar molecule?
What is the molecular shape around the phosphorus atom in PH3? Dibromomethane (CH2Br2) is a nonpolar molecule.
Is CH4 polar or nonpolar electronegativity?
Methane (CH4) is a non-polar hydrocarbon compound composed out of a single carbon atom and 4 hydrogen atoms. Methane is non-polar as the difference in electronegativities between carbon and hydrogen is not great enough to form a polarized chemical bond.
What is the electronegativity of CH4?
However, the values are C = 2.55 and H = 2.20 .
What is the density of Diiodomethane?
3.32 g/cm³
Diiodomethane/Density
What is the molecular geometry of ch2i2?
Starts here2:12CH2Cl2 Molecular Geometry, Bond Angles (and Electron – YouTubeYouTube
Is CH4 polar or non-polar and why?
CH4 is a nonpolar molecule as it has a symmetric tetrahedral geometrical shape with four identical C-H bonds. The electronegativity of carbon and hydrogen is 2.55 and 2.2, respectively, which causes the partial charges to be almost zero. Man-made CH4 mainly comes from the oil and gas industry.
Is if2 polar?
So overall, this is non polar.
What bond is CH4?
covalent bonds
Methane, CH4, is a covalent compound with exactly 5 atoms that are linked by covalent bonds. We draw this covalent bonding as a Lewis structure (see diagram). The lines, or sticks, as we say, represent the covalent bonds.
Is CH2I2 (diiodomethane) polar or nonpolar?
Answer = ch2i2 ( Diiodomethane ) is Nonpolar. What is polar and non-polar? Polar. “In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole or multipole moment. Polar molecules must contain polar bonds due to a difference in electronegativity between the bonded atoms.
Is methane(CH4) polar or nonpolar?
Is Methane (CH4) polar or non-polar? CH4 is a non-polar molecule because it contains four bonds (C-H) that are arranged symmetrically in tetrahedral geometrical shapes. Due to this, dipole moment generated on each side along C-H will cancel out each other making an overall nonpolar molecule.
What is the net dipole moment of CH4?
The net dipole moment in the molecule is the sum of the dipole moments that exist in the molecule. Here as all the dipole moments are canceled out, there is zero dipole moment in the CH4 molecule. And as there is no net dipole moment in the molecule, there are no poles formed for CH4, making methane a nonpolar molecule.
What makes a diatomic molecule nonpolar?
A molecule may be nonpolar either when there is an equal sharing of electrons between the two atoms of a diatomic molecule or because of the symmetrical arrangement of polar bonds in a more complex molecule. (Wikipedia) If the answer is wrong, please comment in below article !