The FDA has granted a breakthrough therapy designation to niraparib (Zejula) for the treatment of men with BRCA1/2-mutant positive metastatic castration-resistant prostate cancer (mCRPC) who have previously received taxane-based chemotherapy and an androgen receptor (AR) inhibitor, according to Janssen, the drug’s …
Which PARP inhibitors are approved for prostate cancer?
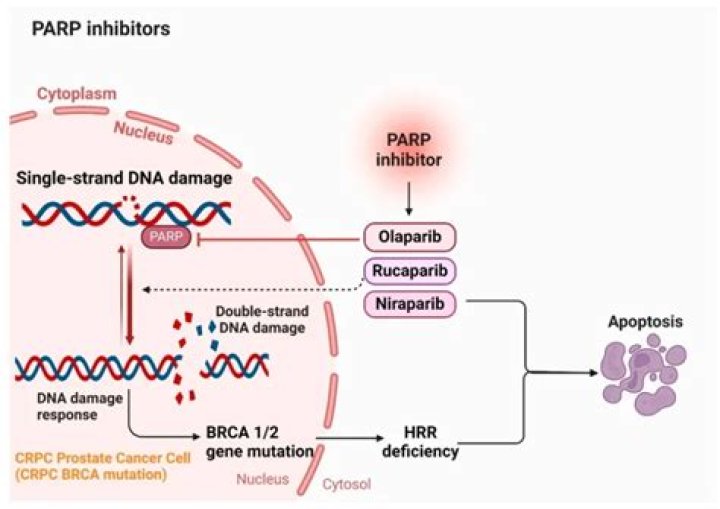
In May 2020, the Food and Drug Administration (FDA) officially approved the first two PARP inhibitors, Rucaparib and Olaparib, for the treatment of prostate cancer (8).
How long do PARP inhibitors work for prostate cancer?
Men treated with the PARP inhibitor went twice as long before experiencing disease progression (7.4 months versus 3.6 months), and early data showed that their overall survival was nearly five months longer than those in the group that received hormonal treatment (19.1 months versus 14.7 months).
How expensive is Niraparib?
Niraparib was the least cost-effective at $18,157/PFS with BRCA mutation and $18,253/PFS without BRCA mutation. Niraparib was the most effective, leading to 29.2 months PFS with mutation and 17.7 months with no mutation. Costs associated with PARP inhibitors were 7.1 to 8.3 times more than platinum combinations.
Is there immunotherapy for prostate cancer?
Immunotherapy for prostate cancer works by helping a patient’s own immune system fight back against cancer cells. One immune therapy that has been approved by the FDA is a vaccine called sipuleucel-T (Provenge), which spurs a patient’s immune system to attack prostate cancer cells.
Which PARP inhibitors are FDA approved?
Four PARPi are currently approved for clinical use: olaparib, rucaparib, niraparib, and talazoparib, with their approvals summarized in the table below.
How effective are PARP inhibitors?
The results indicated that PARP inhibitors significantly improved PFS for ovarian cancer with PFI of >12 months, 6–12 months, and >6 months (HR = 0.39, 95% CI = 0.31–0.48; HR = 0.40, 95% CI = 0.27–0.57; HR = 0.38; 95% CI = 0.32–0.44, respectively).
How effective is niraparib?
For the overall study population, 84% who received niraparib were still alive at 2 years compared with 77% who received placebo. For patients with homologous recombination deficiency, 91% who received niraparib were still alive at 2 years compared with 85% who received placebo.
What is niraparib used for?
Niraparib is used as first-line maintenance treatment in patients with advanced ovarian cancer, fallopian tube cancer, or primary peritoneal cancer. It is also used as maintenance treatment in patients with recurrent ovarian cancer, fallopian tube cancer, or primary peritoneal cancer.
Can immunotherapy raise PSA levels?
The goal is to stimulate your own immune system to fight the cancer cells. This immunotherapy does not lower PSA, treat symptoms, or delay disease progression—however, it has been shown to prolong life.
Is olaparib FDA approved for prostate cancer?
In May 2020, the Food and Drug Administration (FDA) approved two PARP inhibitors for the treatment of prostate cancer. Olaparib was approved for mCRPC patients with any HRR mutation (HRRm; one of 14 genes), who had previously received a second-generation hormonal agent.
What is the difference between olaparib and niraparab?
According to the researchers, these differences could be credited to their selectivity; they cite a recent study in which the data showed niraparib was more selective towards PARP1 and PARP2 while the olaparib and rucaparib were more potent inhibitors of PARP1 but less selective.
How effective is niraparib for progression-free survival?
Five of these studies offered data on progression-free survival (PFS) among patients, throughout which all 3 treatments resulted in statistically significant improved PFS compared with placebo. Compared with placebo, the hazard ratio (HR) for niraparib was 0.6; for rucaparib, 0.7; and for olaparib, 0.72.
Is niraparib better for BRCA mutations?
Specifically looking at PFS among patients with BRCA mutations, data from 4 studies showed that niraparib (HR, 0.69) and olaparib (HR, 0.76) both showed a statistically significant advantage over placebo, while rucaparib did not (HR, 0.79).