2 – Butanol on heating with H2SO4 gives 1 – butene and 2 – butene.
When 2 butanol is heated with concentrated sulfuric acid the main product is?
When butan-2-ol is heated with concentrated H2SO4, but-2-ene is the major product according to Saytzeff rule.
Which alkenes are produced when butan-2-ol is dehydrated with concentrated H2SO4?
Dehydration of butan-2-ol leads to a mixture containing: but-1-ene. cis-but-2-ene (also known as (Z)-but-2-ene) trans-but-2-ene (also known as (E)-but-2-ene)
What is the major product formed when 2 butanol?
2-butene
The formation of 2-butene as a major product by dehydration of 2-butanol is in accordance witha Blanc ruleb Huckel rulec Markownikoffs ruled Saytzeffs rule.
Which of the following is the major product when 1 butanol is heated with conc H2SO4?
Trans-2-butene.
Which of the following is the major product when 1 butanol is heated with concentrated h2so4?
Which of the following is the major product when 1 butanol is heated?
trans- 2 – butene.
When butan-2-ol is dehydrated using Sulphuric acid?
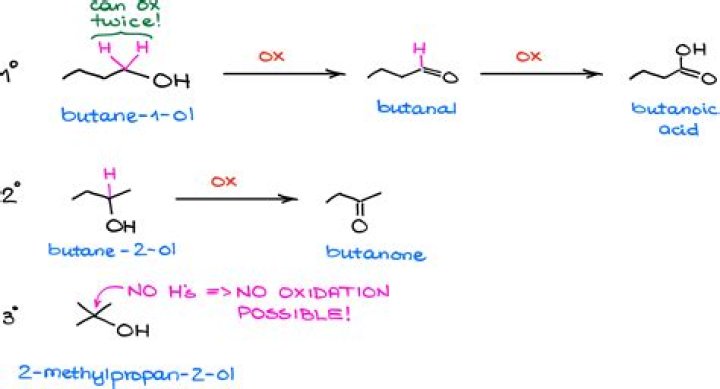
When Butan-2-ol is dehydrated using sulphuric acid, the concentration of acid and temperature needed respectively is 60% cone. and 373 K. Explanation: Secondary alcohol (2°) is dehydrated by heating with 60% H2SO4 at 373 K.
When excess 2-butanol is heated with concentrated Sulphuric acid at 180 C the major product is?
trans- 2 – butene
trans- 2 – butene.
When 1 butanol is heated in the presence of concentrated sulfuric acid what compound is formed?
But-2-ene is obtained as the major product. First n-butyl (1∘) carbocation is formed .
What happens when 2 – butanol is heated with H2SO4?
Q. Assertion : 2 – Butanol on heating with H 2 S O 4 gives 1 – butene and 2 – butene. Reason : Dehydration of 2 – butanol follows Saytzeff’s rule. Saytzeff’s rule: The alkene formed in greatest amount is the one that corresponds to removal of the hydrogen from the β -carbon having the fewest hydrogen substituent.
How many alkenes are formed when butan2ol is dehydrated?
Butan-2-ol is a good example of this, with no less than three different alkenes being formed when it is dehydrated. To make the diagrams less cluttered, we’ll use the simplified version of the mechanism showing gain and loss of H+.
What is the reaction between sulphuric acid and alcohol?
In the first stage, the alcohol is protonated by picking up a hydrogen ion from the sulphuric acid. In the second stage, the positive ion then sheds a water molecule and produces a carbocation.
Which alkene is formed in the greatest amount of hydrogen?
Saytzeff’s rule: The alkene formed in greatest amount is the one that corresponds to removal of the hydrogen from the β -carbon having the fewest hydrogen substituent. In case of 2 ∘ and 3 ∘ alc. Saytzefi’s rule is followed