6g/litre.
What does a 20 volume hydrogen peroxide refer?
I buy it often. So, it would seem intuitive that “20 volume Hydrogen Peroxide” would be a 6% solution. To check, let’s take 1 liter of H2O2 and let it decompose: 20 liters of O2 is 20/22.4 = 0.893 moles of O2. To get 0.893 moles of O2, you must have started with 2 x 0.893 = 1.786 moles of H2O2, which weighs 60.7 grams.
What do you mean by 20 volume of hydrogen peroxide calculate the strength of 20 volume of hydrogen peroxide?
A solution of hydrogen peroxide labelled as 20 volume actually means that 1 ml of hydrogen peroxide solution on decomposition by heat produces 20 ml of oxygen at NTP. Let us now calculate the strength of 20 volume H2O2 in gram/litre.
What does 20v H2O2 mean?
20VH2O2 means that 1 litre of H2O2 solution would produce 20 litre of Oxygen.
What percentage is 20 vol?
Why Developer Strengths Matter Hydrogen peroxide or developer strength is typically measured in volumes of oxygen liberated per volume of solution. The strength may also use percentages to indicate the percentage of peroxide. For example: 3% = 10 volume, 6% = 20 volume, 9% = 30 volume and 12% = 40 volume.
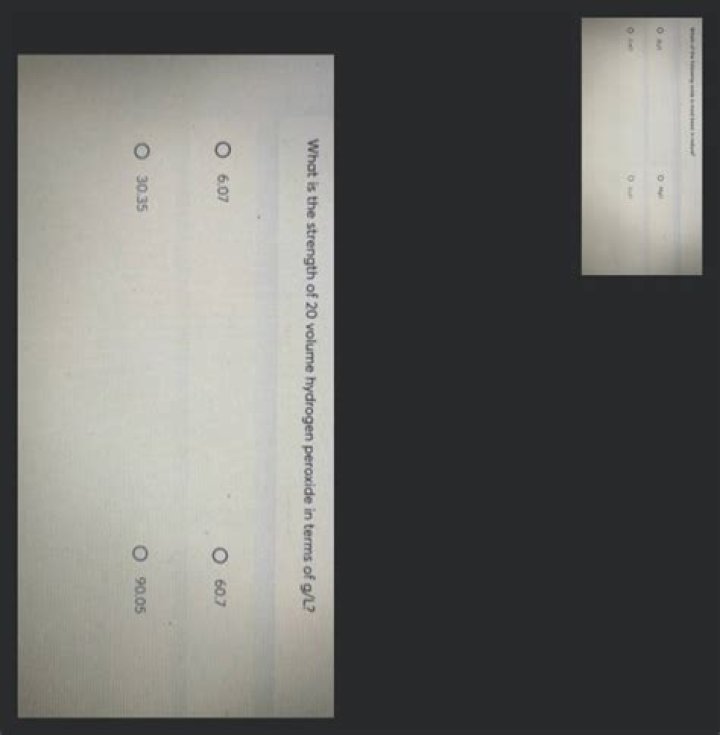
What is the strength of 20 volume hydrogen peroxide in terms of g Litre?
6gL−1.
How do you calculate percentage of hydrogen peroxide?
So, there would be 31.998 grams per mole of oxygen in hydrogen peroxide. We divide that by the total mass of the compound, multiply it by 100%, and get the mass percent of oxygen in hydrogen peroxide being about 94%.
What is the normality of 20 volume hydrogen peroxide solution?
To calculate the strength in g/l of 20 volume H2O2 solution. By definition, 1 litre of 20 volume H2O2 solution on decomposition gives 20 litres of oxygen at N.T.P. Consider the chemical equation, 2H2O22×34=68g→2H2O+O222.4 Litres at N.T.P.
What is the concentration of 20 volume hydrogen peroxide in gram Litre?
How do you find the percent strength of hydrogen peroxide?
Strength of hydrogen peroxide: (i) As percentage. It is expressed as W/V percentage of H2O2 in solution. Thus, 30% solution of H2O2 means 30 grams of H2O2 are present in 100 ml of solution.
What percentage of hydrogen peroxide is hydrogen?
5.926%
So the mass of hydrogen in hydrogen peroxide is 2.0158 grams per mole, because there’s two hydrogens. We have to divide that by the molar mass of the entire compound. Multiply that by 100%, and we see that the mass percent of hydrogen in hydrogen peroxide is 5.926%.
What is 10 volume peroxide used for?
10 Volume Peroxide is a standard oxidizing strength for permanent, no-lift haircolor . Designed for use when you simply want to add a tint or color tone to hair of the same lightness level, 10 Volume Peroxide opens the cuticle layer of the hair allowing the color molecules to penetrate and color to be deposited in the cortex.
What is the equation for hydrogen peroxide?
Hydrogen peroxide is a chemical substance very used as bleach, oxidizing and anti-microbial agent. Formula and structure: Hydrogen peroxide has the chemical formula H2O2. Its molecular formula is H2O2 and its molar mass is 34.0147 g mol-1.
Is hydrogen peroxide a weak acid?
Hydrogen peroxide is a weak acid, and it can form hydroperoxide or peroxide salts or derivatives of many metals. For example, on addition to an aqueous solution of chromic acid ( CrO3 ) or acidic solutions of dichromate salts, it will form an unstable blue peroxide CrO(O2)2.
What is the breakdown of hydrogen peroxide?
The breakdown of hydrogen peroxide into water and oxygen is catalysed by catalase enzymes in plants and animals. Hydrogen peroxide naturally breaks down to water and oxygen, but very slowly. The catalase enzyme in yeast and other materials, such as potato and liver, speeds up the reaction greatly.